Abstract
Comparative analysis and molecular characterization of genomic sequences and proteins of FABP4 and FABP5 from the giant panda (Ailuropoda melanoleuca)
Author(s): B. Song, Y.L. Hou, X. Ding, T. Wang, F. Wang, J.C. Zhong, T. Xu, J. Zhong, W.R. Hou and S.R. ShuaiFatty acid binding proteins (FABPs) are a family of small, highly conserved cytoplasmic proteins that bind long-chain fatty acids and other hydrophobic ligands. In this study, cDNA and genomic sequences of FABP4 and FABP5 were cloned successfully from the giant panda (Ailuropoda melanoleuca) using reverse transcription polymerase chain reaction (RT-PCR) technology and touchdown-PCR. The cDNAs of FABP4 and FABP5 cloned from the giant panda were 400 and 413 bp in length, containing an open reading frame of 399 and 408 bp, encoding 132 and 135 amino acids, respectively. The genomic sequences of FABP4 and FABP5 were 3976 and 3962 bp, respectively, which each contained four exons and three introns. Sequence alignment indicated a high degree of homologywith reported FABP sequences of other mammals at both the amino acid and DNA levels. Topology prediction revealed seven protein kinase C phosphorylation sites, two casein kinase II phosphorylation sites, two N-myristoylation sites, and one cytosolic fatty acid-binding protein signature in the FABP4 protein, and three N-glycosylation sites, three protein kinase C phosphorylation sites, one casein kinase II phosphorylation site, one N-myristoylation site, one amidation site, and one cytosolic fatty acid-binding protein signature in the FABP5 protein. The FABP4 and FABP5 genes were overexpressed in Escherichia coli BL21 and they produced the expected 16.8- and 17.0-kDa polypeptides. The results obtained in this study provide information for further in-depth research of this system, which has great value of both theoretical and practical significance.
Impact Factor an Index

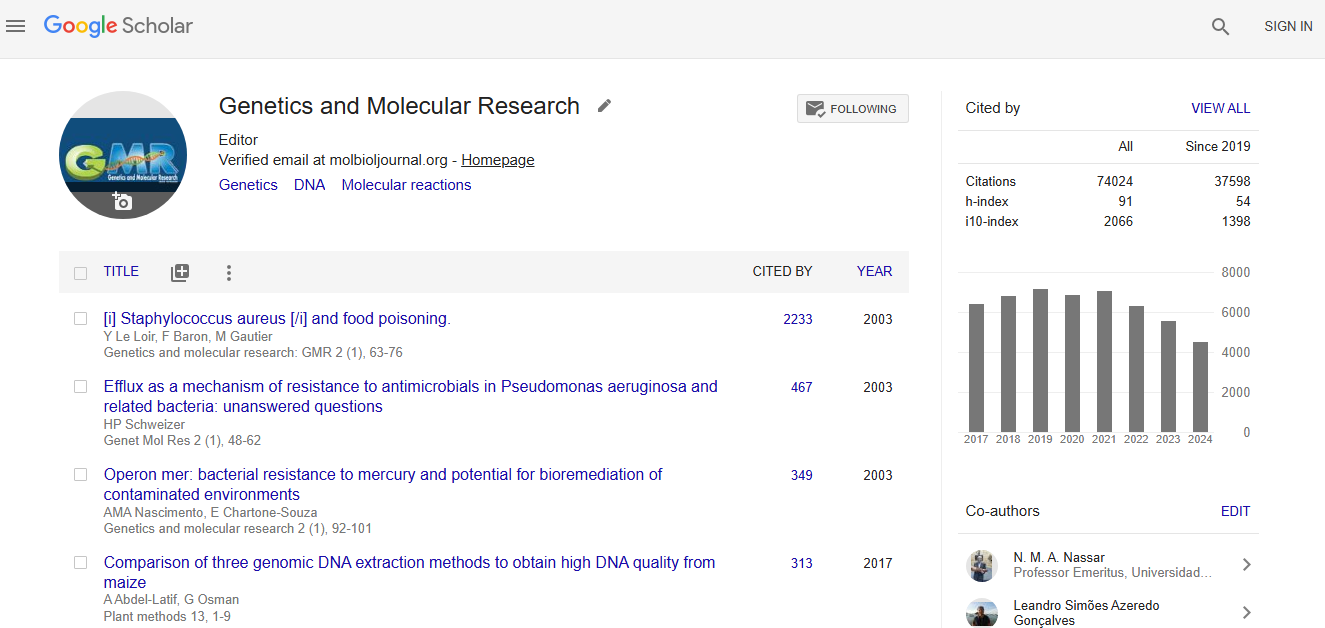
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report