Abstract
Intracellular localization and association of MHC class I with porcine invariant chain
Author(s): F.Z. Xu S.G. Wu W.Y. YuThe objective was to investigate the intracellular localization and association of pig major histocompatibility complex (MHC) class I subunits with invariant chain (Ii). Pig MHC class I subunit cDNAs were cloned by RT-PCR and eukaryotic expression plasmids of �?± and �?²2m were constructed with fusions to red or enhanced green fluorescent protein (pDsRed2-N1-�?±, pEGFP-N1-�?±, pDsRed2-N1-�?²2m, and pEGFP-N1-�?²2m). A pig Ii mutant with a deleted CLIP region (DCLIP-Ii) was constructed by overlap extension PCR. Wild-type Ii and mutant Ii were cloned into pEGFP-C1 (pEGFP-C1-Ii, pEGFP-C1-DCLIP-Ii). The recombinant plasmids of MHC I subunits and pEGFP-C1-Ii (pEGFP-C1-DCLIP-Ii) were transiently cotransfected into COS-7 cells with Lipofectamine 2000. Immunofluorescence microscopy was performed to detect expression and intracellular localization of Ii and MHC I subunits, and immunoprecipitation was used to analyze their association. Our results indicated that pig Ii associates with integrated MHC I subunits to form oligomers, but cannot associate with single MHC I subunits. Furthermore, deletion of the Ii CLIP sequence blocks association with integrated MHC I subunits. Thus, pig Ii cannot associate with a single MHC I molecule, the �?± or �?²2m chain, but Ii and the integrated MHC I molecule can form complexes that colocalize in the endomembranes of COS-7 cells. The Ii of CLIP plays a key role in assembly of Ii and MHC I. The objective was to investigate the intracellular localization and association of pig major histocompatibility complex (MHC) class I subunits with invariant chain (Ii). Pig MHC class I subunit cDNAs were cloned by RT-PCR and eukaryotic expression plasmids of �?± and �?²2m were constructed with fusions to red or enhanced green fluorescent protein (pDsRed2-N1-�?±, pEGFP-N1-�?±, pDsRed2-N1-�?²2m, and pEGFP-N1-�?²2m). A pig Ii mutant with a deleted CLIP region (DCLIP-Ii) was constructed by overlap extension PCR. Wild-type Ii and mutant Ii were cloned into pEGFP-C1 (pEGFP-C1-Ii, pEGFP-C1-DCLIP-Ii). The recombinant plasmids of MHC I subunits and pEGFP-C1-Ii (pEGFP-C1-DCLIP-Ii) were transiently cotransfected into COS-7 cells with Lipofectamine 2000. Immunofluorescence microscopy was performed to detect expression and intracellular localization of Ii and MHC I subunits, and immunoprecipitation was used to analyze their association. Our results indicated that pig Ii associates with integrated MHC I subunits to form oligomers, but cannot associate with single MHC I subunits. Furthermore, deletion of the Ii CLIP sequence blocks association with integrated MHC I subunits. Thus, pig Ii cannot associate with a single MHC I molecule, the �?± or �?²2m chain, but Ii and the integrated MHC I molecule can form complexes that colocalize in the endomembranes of COS-7 cells. The Ii of CLIP plays a key role in assembly of Ii and MHC I.
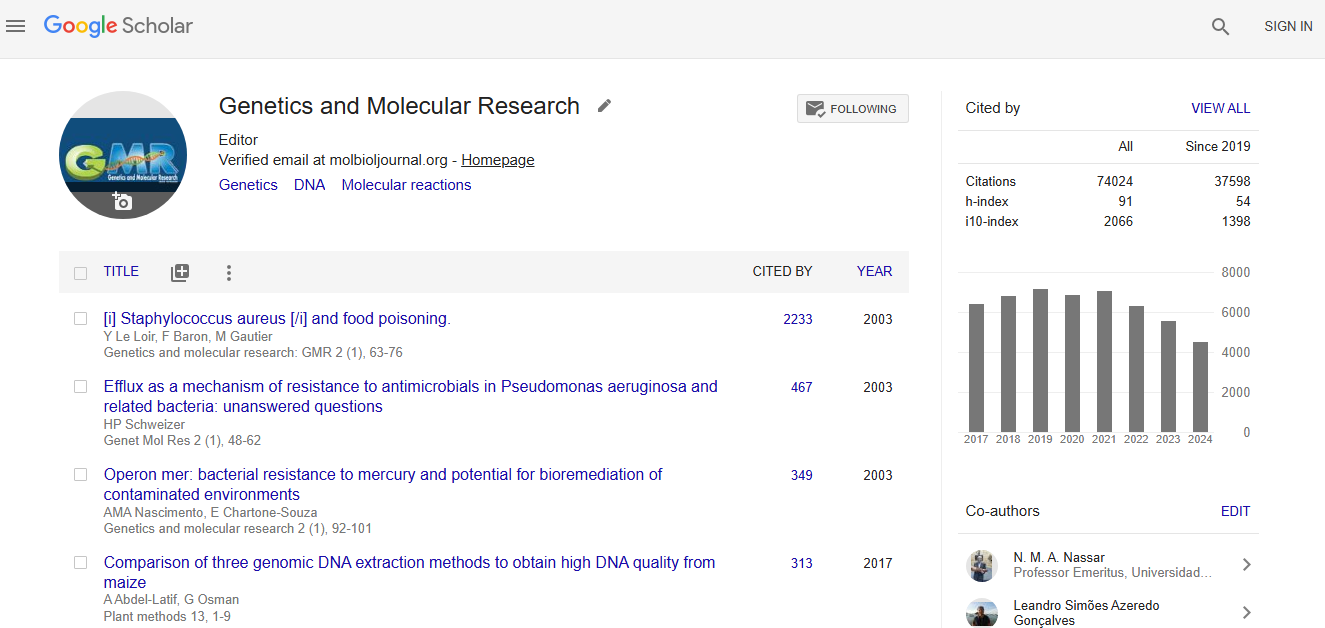
Impact Factor an Index

Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report