Abstract
Molecular cloning and characterization of a subtilisin-like protease from Arabidopsis thaliana
Author(s): D.H. Li, H. Xi, X.B. Yu and Y.P. CaiThe Arabidopsis thaliana genome encodes 56 subtilisin-like serine proteases (subtilases). In order to evaluate the protease activity of a previously uncharacterized subtilase, designated as AtSBT1.9, we cloned its full-length cDNA from A. thaliana seedlings. An AtSBT1.9 mature peptide coding sequence was inserted into the bacterial expression vector, pMAL-c2x, and the recombinant vector was transformed into Escherichia coli BL21 (DE3). The recombinant AtSBT1.9 tagged by maltose binding protein (MBP) was induced as a 117.5-kDa protein in the soluble form in E. coli BL21 (DE3). MBP-AtSBT1.9 was expressed at a level of 11% (w/w) of the bacterial total protein. Protein purification using Amylose Resin revealed a recombinant AtSBT1.9 protease activity of 9.23 U/mg protein at pH 7 and 25°C. Maximal activity occurred over a broad pH (7-8) and temperature (25°-42°C) optimal range. Validation of AtSBT1.9 protease activity would help in characterizing its in vivo function in A. thaliana.
Impact Factor an Index

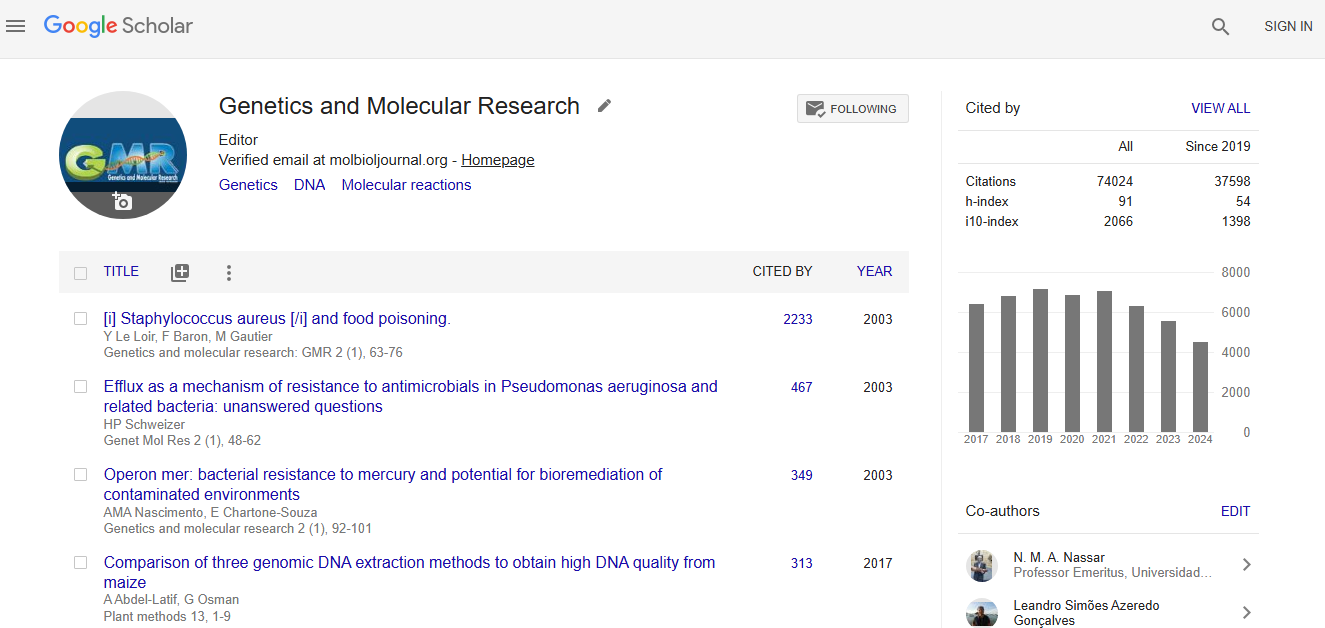
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report