Abstract
Rapid and sensitive LC-MS/MS method for the determination of auraptene in rat plasma and its application in a pharmacokinetic and bioavailability study in rats
Author(s): X.D. Ye, H. Ouyang, L.Y. Zhong, T.E. Li, X.Y. Rao, Y.L. Feng and W.L. YangA simple, sensitive and specific liquid chromatography-tandem mass spectrometry method was developed and validated for the determination of auraptene, a constituent isolated from Fructus aurantii with potential to combat Alzheimer’s disease, in rat plasma. Rat plasma samples were pretreated by protein precipitation with methanol. The analytes were separated by a Waters Sun Fire C18 column (50 mm x 2 mm, 5 μm) and eluted with 1:1000 methanol and formic acid/water (v/v) mobile phase with a flow rate of 0.5 mL/min. Multiple reaction monitoring was used to monitor the transition of the deprotonated auraptene molecule with an m/z of 299.3 [M+H]+, to the product ion with an m/z of 162.9 [M+H]+. Progesterone, with an m/z of 315.2→ 96.9 was used as an internal standard. The limits of detection and of quantification of auraptene in the rat plasma were 1 and 5 ng/mL, respectively. The method was linear in the concentration range of 20- 2000 ng/mL with coefficient correlation of 0.9956. After auraptene (100 mg/kg, p.o.) administration, the maximum plasma concentration and the time taken to reach maximum concentration were 1719.5 ± 384.3 g/mL and 108.0 ± 25.3 min, respectively. The elimination half-life was 108.0 ± 25.3 for auraptene (100 mg/kg, p.o.) and 3.0 ± 0 min for auraptene (2 mg/kg, i.v.). The oral bioavailability was about 8.5%.
Impact Factor an Index

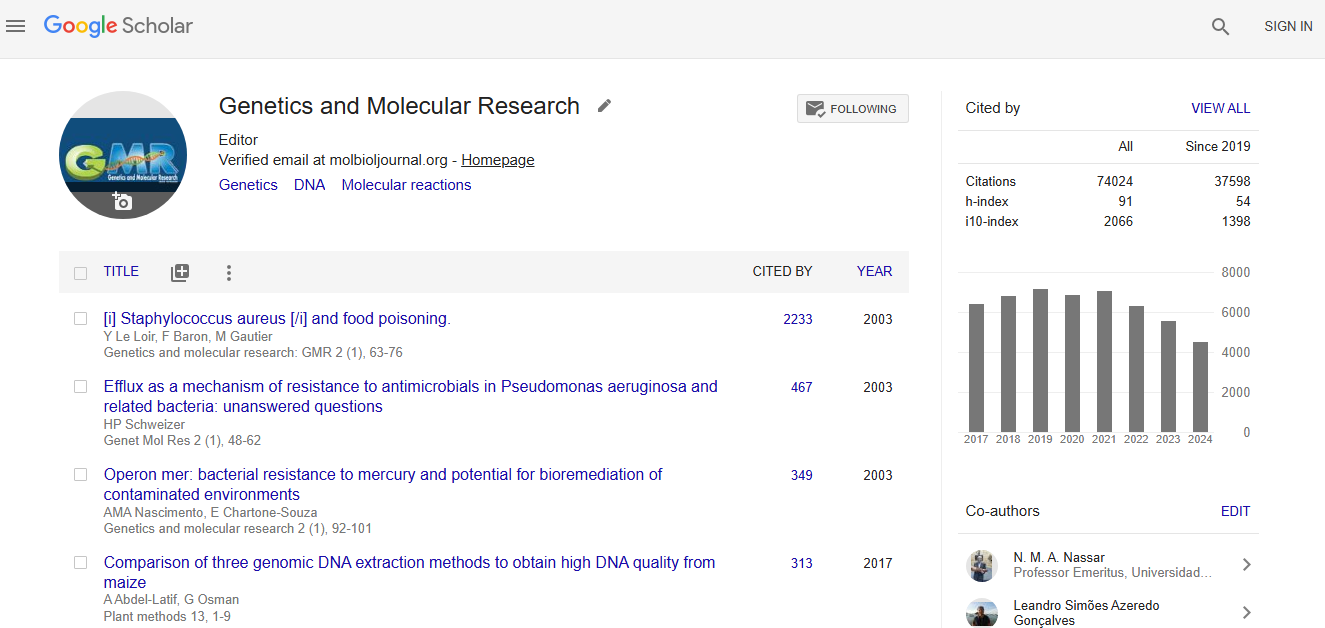
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report